For broad coordinated access to patient-related lung research data for scientific purposes, study data is stored centrally in the DZL Data Warehouse (DZL DWH) and is kept confidential (pseudonymous). The basis for this is always a corresponding patient consent, which can be revoked at any time. With the DZL Broad Consent, patient data can be acquired based on a DZL wide harmonized legal and organizational standard.
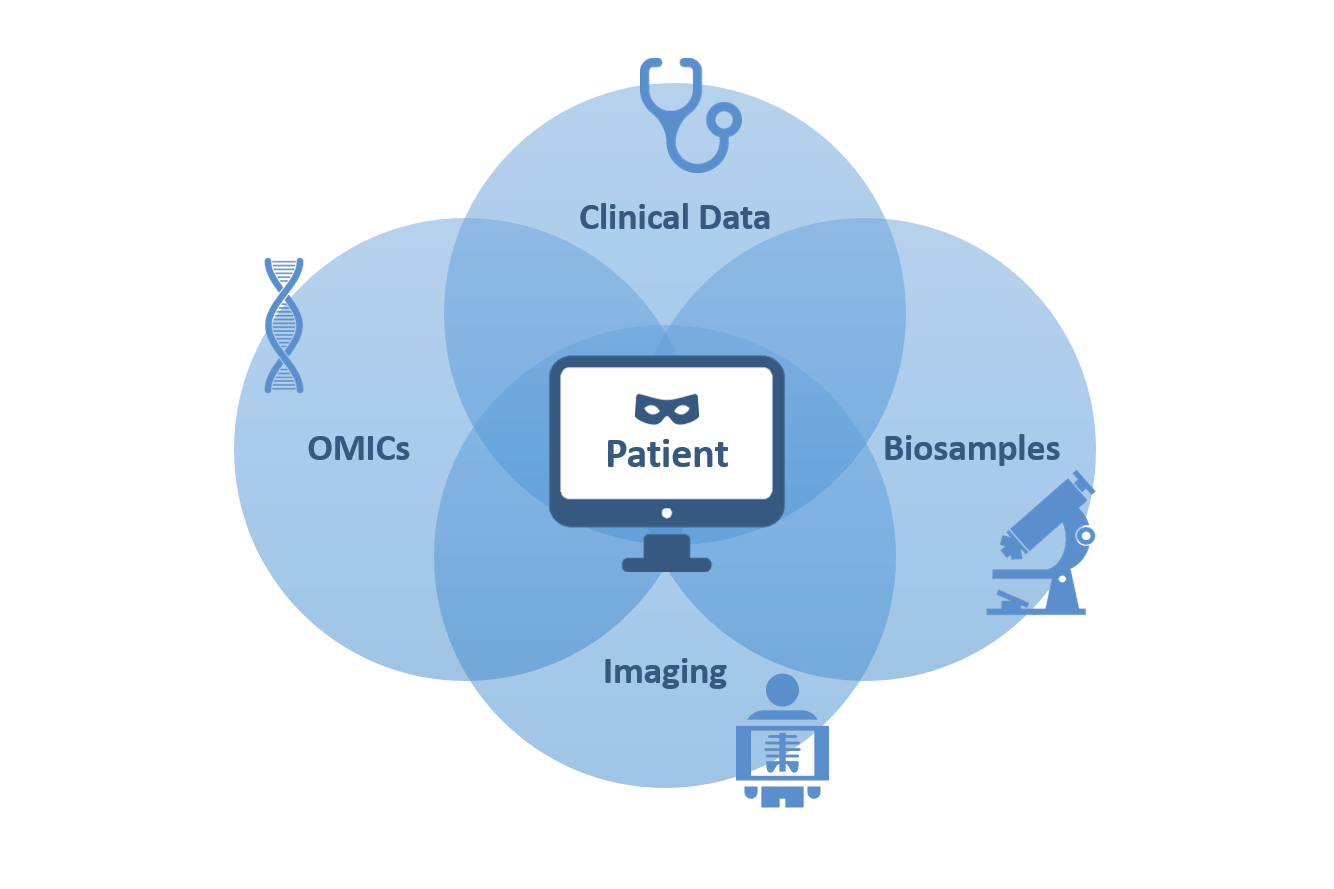
The Rules of Procedure for Sample and Data Processing (GOPDV) in the DZL regulates the handling of clinical and patient-derived research data within the DZL. DZL members, their cooperation partners and external researchers can use the data contained according to a regulated procedure. The ownership of the data remains with the local data-collecting agencies and biobanks as well as (image) databases.
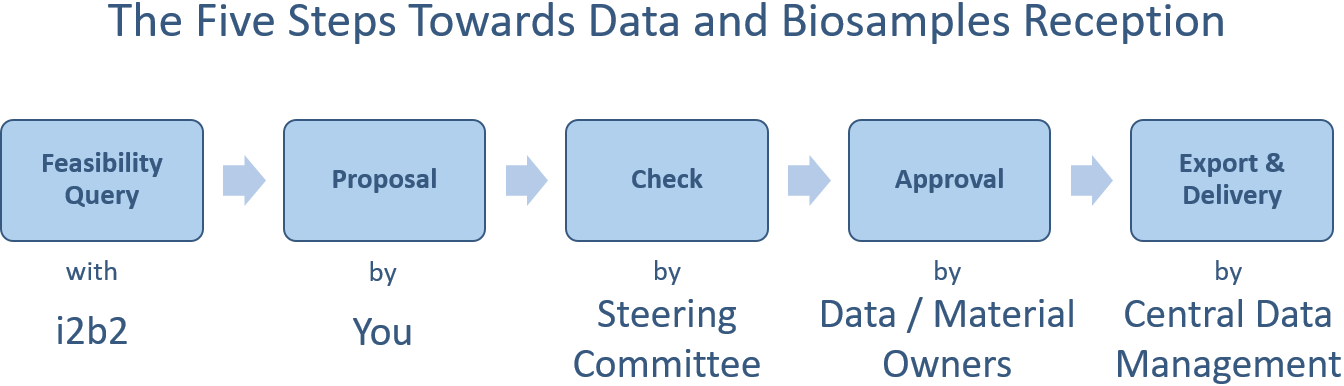
The use of (image) data and biomaterials from the DZL DWH must be requested upon written application. Basic requirements for the approval of an application are a defined scientific project sketch and the valid vote of the local ethics committee. The steering committee decides on the approval and procedure in a multi-stage process (see GOPDV § 3 (3) and Appendix 1). The DZL Ethics Panel provides ethical advice on the use of (image) data and samples.
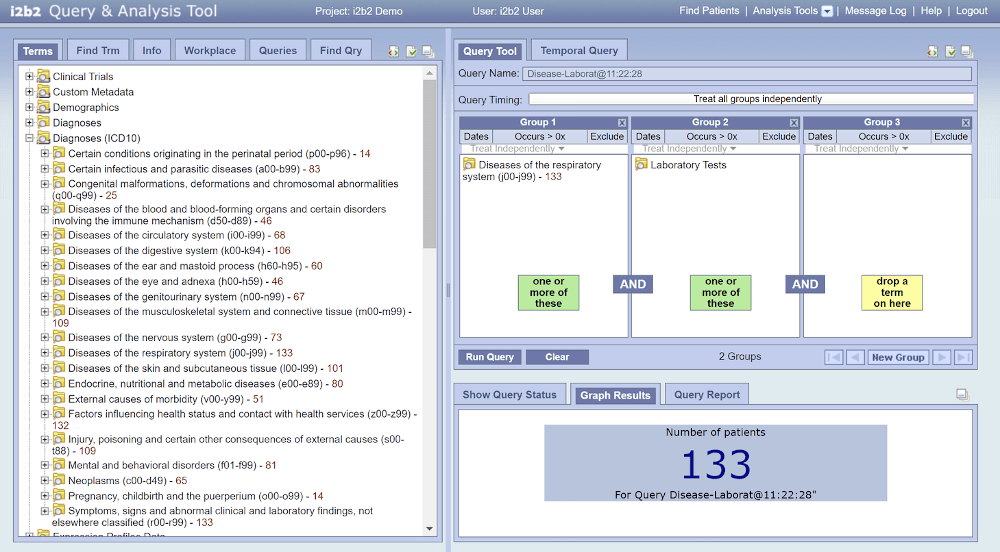
The open source software i2b2 (Informatics for Integrating Biology and the Bedside) developed by Harvard University and MIT is used as software solution for the DZL Central Data Warehouse. It allows feasibility studies through queries about existence and number of patient records by using logical combinations and filters of clinical parameters. Access is provided through the DZL Data Warehouse online portal. All PIs of the DZL already have an i2b2 access. Temporary access can be given upon request to the Central Data Management. All hardware and software components as well as data are operated within Germany.

Combining the data from different lung centers requires a common ontology that contains the essential information such as phenotypes, measured data and sample data in structured form. This includes various annotations such as clear definitions and references to corresponding elements of other classifications such as LOINC or SNOMED-CT. DZL Data Management develops these with the help of the expertise of all sites and disease areas. The metadata management is implemented in the Collaborative Metadata Repository (CoMetaR) as platform for creating, editing, and visualizing a common ontology. The lung reasearch specific concept tree including annotations can be viewed and browsed via the DZL CoMetaR online portal. Trained personnel, such as medical documentaries, can also participate in the editing process. If you are interested, please contact the DZL Central Data Management.